India has become a global hub for high-quality and affordable medicines, earning its reputation as the “pharmacy of the world.” For businesses, distributors, and healthcare providers across the globe, importing pharmaceutical products from India offers a reliable way to access cost-effective medicines without compromising on quality. However, entering the Indian pharmaceutical market requires a clear understanding of its regulatory system, documentation, and compliance standards.
Importing pharmaceutical medicines from India in 2026 means following strict rules set by the Central Drugs Standard Control Organisation (CDSCO). These rules include getting an import license (Form 10), registering products using Forms 41/40, and ensuring that all medicines meet WHO-GMP quality standards. Important steps also include appointing an authorised agent in India, applying through the SUGAM online portal, and maintaining proper cold-chain systems for medicines that need controlled temperatures.
This ultimate guide for 2026 is designed to simplify the entire process for international buyers. Whether you are a distributor, hospital supplier, or pharma entrepreneur, this guide will help you navigate regulations, avoid common mistakes, and build a smooth, compliant import process from India.
Steps To Importing Pharmaceutical Medicines From India
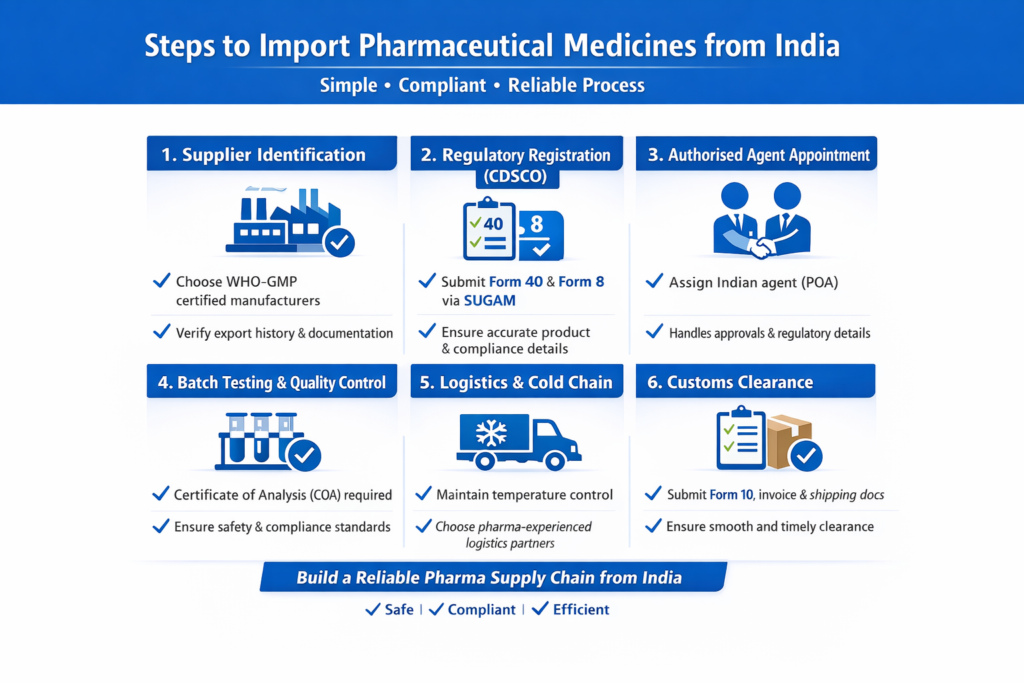
Understanding the steps to import medicines from India is essential for ensuring a smooth, compliant, and risk-free process. Below is a detailed breakdown of how to import medicines from India in a structured and practical way:
1. Supplier Identification
The first step in the process of importing pharmaceuticals from India is selecting a reliable and certified supplier. Look for manufacturers or exporters who are WHO-GMP certified, have a strong export history, and can provide complete documentation. It’s important to verify their credentials, production capacity, and product quality before entering into any agreement. Conducting due diligence at this stage helps prevent future compliance or quality issues.
2. Regulatory Registration (CDSCO)
A critical part of how to import medicines from India is completing regulatory registration with the Central Drugs Standard Control Organisation (CDSCO). This involves submitting Form 40 for product registration and Form 8 for obtaining an import license through the SUGAM portal. Proper documentation, including product details, manufacturing information, and certifications, must be submitted accurately to avoid delays or rejections.
3. Appointment of Authorised Agent
One of the mandatory steps to import medicines from India is appointing an authorised Indian agent through a Power of Attorney (POA). This agent acts as your official representative in India and handles regulatory submissions, approvals, and communication with CDSCO. A knowledgeable agent ensures that all processes are handled efficiently and in compliance with Indian regulations.
4. Batch Testing & Quality Control
Quality assurance is a crucial stage in the steps to import pharmaceuticals from India. Each batch of medicines must be accompanied by a Certificate of Analysis (COA) to confirm quality and composition. In some cases, additional testing may be required by Indian authorities or independent labs to ensure safety, stability, and compliance with standards.
5. Logistics and Cold Chain Management
Efficient logistics planning is key to how to import medicines from India, especially for temperature-sensitive products such as vaccines, biologics, or injectables. Maintaining a proper cold chain during storage and transportation ensures product integrity. Choosing experienced logistics partners with pharma handling expertise reduces the risk of damage or spoilage.
6. Customs Clearance
The final step in the process of importing medicines from India is customs clearance. This involves submitting essential documents such as the Import License (Form 10), Bill of Entry, invoice, packing list, and shipping documents. Proper coordination with customs authorities ensures timely clearance and avoids unnecessary delays or penalties.
By carefully following these steps to import pharmaceuticals from India, importers can streamline operations, maintain compliance, and build a reliable supply chain for long-term success.
Regulatory Framework & Licensing (2026)
Importing pharmaceutical medicines from India requires strict compliance with regulations set by the Central Drugs Standard Control Organisation (CDSCO). This authority ensures that all imported products meet the required safety, quality, and efficacy standards before they enter the Indian market. Understanding the regulatory structure is essential for avoiding delays, penalties, or rejection of applications.
CDSCO Registration (Form 41)
One of the essential steps to import pharmaceuticals from India is registering both the manufacturing site and the products with CDSCO. This is done by obtaining a Registration Certificate (Form 41). The process involves submitting detailed documentation, including manufacturing practices, product composition, stability data, and quality certifications such as WHO-GMP. This step confirms that the foreign manufacturer meets Indian regulatory standards.
Import License (Form 10)
Once registration is approved, the next step is to apply for an Import License using Form 8 (or Form 8A for Schedule X drugs) through the SUGAM online portal. The license (issued in Form 10) allows the legal import of pharmaceutical products into India. Each product and variation may require separate approval, so accuracy in documentation is critical.
Appointment of an Authorised Agent
Another important part of importing pharmaceutical medicines and products from India is appointing an authorised agent. Foreign manufacturers cannot directly handle regulatory procedures in India. They must appoint an authorised Indian agent who acts as the official representative. This agent is responsible for submitting applications, coordinating with CDSCO, handling compliance requirements, and managing communication with authorities. Choosing a reliable and experienced agent is crucial for smooth approval and long-term regulatory compliance.
By following these key steps to import medicines from India, importers can ensure a legally compliant and efficient entry into the Indian pharmaceutical market.
Key Documentation Required
Certificate of Pharmaceutical Product (COPP) – Validates the product is sold in India.
WHO GMP Certificate – Proof of Good Manufacturing Practices.
Form 9 & 40 – Undertakings and application forms.
Technical Documents – Drug Master File (DMF), Certificate of Analysis (COA), and Certificate of Origin.
Power of Attorney – Must be apostilled or authenticated if the manufacturer is not the importer.
Final Words
Importing medicines from India is a great opportunity for global businesses, but it requires proper planning and compliance. Understanding how to import medicines from India means following the right regulations, documentation, and quality standards at every step.
By following the correct steps to import medicines from India and complying with guidelines set by the Central Drugs Standard Control Organisation (CDSCO), you can ensure a smooth and risk-free process. From supplier selection to logistics and customs clearance, each stage is important.
With the right approach and by following the key steps to import pharmaceuticals from India, you can build a reliable and successful pharmaceutical supply chain.



